Let Uncle Ralph educate you
Synthetic Viral Genomics:
Risks and Benefits for Science and Society
These enzymes can be used to create unique interconnecting junctions, which can be subsequently removed from final assembly product allowing seamless reconstruction of an exact sequence

The pathogenicity of these chimeric coronaviruses is unknown
The pathogenicity of these chimeric coronaviruses is unknown
The pathogenicity of these chimeric coronaviruses is unknown
This powerful technique provides bioterrorists with a “scapegoat” option; leaving a sequence signature that misdirects efforts at tracking the true originators of the crime. Even better, the approach could be used to build mistrust &/or precipitate open warfare

Hence Ecohealth DARPA/DTRA spooks & virus thieves collaboration with Baric (UNC) Lipkin (Mailman) NIchols (Atlanta CDC) and USAMRIID (Bavari, Totura et al) & Jonathan Epstein's palpable concern about dual use references in the @USRightToKnow FOIA emails

Delving into the cold and calculating mind of a twisted genius?
https://t.co/MrwxYK93cx
Bring Uncle Ralph and his transgenic mice in for questioning!
unroll @threadreaderapp

https://t.co/rjNZ8aDNDl

1. Synthetic Genomics: Options for Governance (2008) https://t.co/A32jEfL1TQ
2. Sequence Screening - Robert Jones (2005)
https://t.co/zOqMroYbVU
3. Synthetic Biology as a Field of Dual-Use Bioethical Concern - Alexander Kelle
https://t.co/Va1movPqvs
4. Sanghvi Y. A Roadmap to the Assembly of Synthetic DNA from Raw Materials.
https://t.co/4kWu4vzcXC
5. Collett MS. Impact of Synthetic Genomics on the Threat of Bioterrorism with Viral Agents.
https://t.co/OcaTmuCUBy
6. Fleming DO. Risk Assessment of Synthetic Genomics: A Biosafety & Biosecurity Perspective.
https://t.co/JBcLVLr005
7. Risk Governance of Synthetic Biology
https://t.co/9BE9X5s0Fb
8. US Competitiveness in Synthetic Biology
https://t.co/OCZJdsXSjl
9. Ensuring security of synthetic biology
https://t.co/7kPE1OpbrS
10. Synthetic biology: emerging research field in China
https://t.co/T3BcaDvUsa
11.
What rough beast? Synthetic biology, uncertainty,& the future of biosecurity (2016)
https://t.co/XTlBXILEWh

Reverse genetics with a full-length infectious cDNA of severe acute respiratory syndrome coronavirus (2003)
https://t.co/k8PI1rbcm8
Systematic Assembly of a Full-Length Infectious Clone of Human Coronavirus NL63 (2008)
https://t.co/bWhAv1wsC0
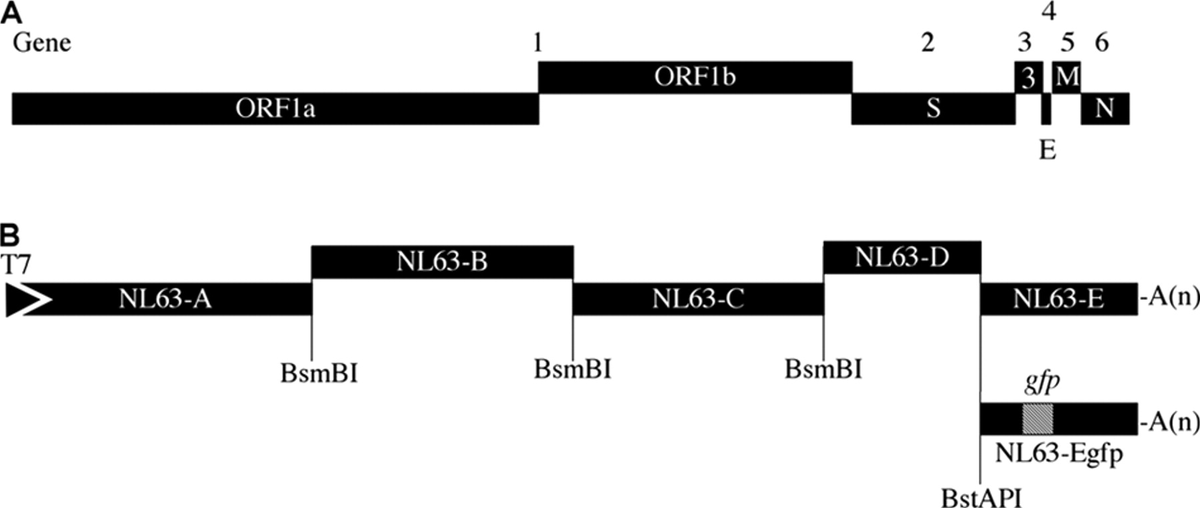
In 2016, Shi and her team at the WIV, in conjunction with the New York-based EcoHealth Alliance, constructed a full-length clone of a bat coronavirus called SL-CoV WIV1. They assembled it in discrete segments.
“Thus, several options exist to remove the desired insert DNA with a single restriction digestion.”
This shows that researchers at the WIV have the ability to genetically engineer viruses and remove the signatures of the genetic engineering.
They showed how they can insert new spikes into viruses. The researchers state:
“Then any spike could be substituted into the genome of SARSr-CoV WIV1 through this strategy.”
COVID 19: The Spike and the Furin Cleavage
https://t.co/bDOMmhpJ1t
More from Billy Bostickson 🏴👁&👁 🆓
Coronavirus nsp10/nsp16 Methyltransferase Can Be Targeted by nsp10-Derived Peptide In Vitro and In Vivo To Increase or Decrease Replication and Pathogenesis
https://t.co/YMlsqFOvZp

Deyin Guo 2016:
Molecular mechanisms of coronavirus RNA capping and methylation
https://t.co/xdEU5goLB6
Deyin Guo 2012
Short peptides derived from the interaction domain of SARS coronavirus nonstructural protein nsp10 can suppress the 2′-O-methyltransferase activity of nsp10/nsp16 complex
https://t.co/x30jPYFDTa

Deyin Guo 2013
Structure-function Analysis of SARS Coronavirus RNA Cap Guanine-N7 Methyltransferase.
https://t.co/NG57b85hcs

Deyin Guo 2016
Identification and Characterization of a Ribose 2’-O-Methyltransferase Encoded by the Ronivirus Branch of Nidovirales
https://t.co/zZ5V7erseD

@NBA @StephenKissler @yhgrad 1. From Day 1, SARS-COV-2 was very well adapted to humans .....and transgenic hACE2 Mice
1. From Day 1, SARS-COV-2 was very well adapted to humans .....and transgenic hACE2 Mice
— Billy Bostickson \U0001f3f4\U0001f441&\U0001f441 \U0001f193 (@BillyBostickson) January 30, 2021
"we generated a mouse model expressing hACE2 by using CRISPR/Cas9 knockin technology. In comparison with wild-type C57BL/6 mice, both young & aged hACE2 mice sustained high viral loads... pic.twitter.com/j94XtSkscj
@NBA @StephenKissler @yhgrad 2. High Probability of serial passaging in Transgenic Mice expressing hACE2 in genesis of SARS-COV-2
1. High Probability of serial passaging in Transgenic Mice expressing hACE2 in genesis of SARS-COV-2!
— Billy Bostickson \U0001f3f4\U0001f441&\U0001f441 \U0001f193 (@BillyBostickson) January 2, 2021
2 papers:
Human\u2013viral molecular mimicryhttps://t.co/irfH0Zgrve
Molecular Mimicryhttps://t.co/yLQoUtfS6s https://t.co/lsCv2iMEQz
@NBA @StephenKissler @yhgrad B.1.1.7 has an unusually large number of genetic changes, ... found to date in mouse-adapted SARS-CoV2 and is also seen in ferret infections.
https://t.co/9Z4oJmkcKj

@NBA @StephenKissler @yhgrad We adapted a clinical isolate of SARS-CoV-2 by serial passaging in the ... Thus, this mouse-adapted strain and associated challenge model should be ... (B) SARS-CoV-2 genomic RNA loads in mouse lung homogenates at P0 to P6.
https://t.co/I90OOCJg7o

dsRNA viruses molecular biology
https://t.co/lwrQoo6ygG

2. Emerging Viruses Group
https://t.co/ND56gVGOAn
3. Structural Biology of Viral Genome Replication
https://t.co/wUqc0YIP3X
4. Molecular Biology of hepatitis Viruses & Gene Therapy
https://t.co/snBbxkhNsC
5. Insect Virus Genetic Engineering Lab
https://t.co/S68mVOA2Ob
h/t @DrKevinWMcCair1 video:
https://t.co/1lrFeWcakB
From Daily Expose:
U.S. DoD awarded a contract for ‘COVID-19 Research’ in Ukraine 3 months before Covid was known to even
2. The findings seem to be kosher and can "currently" be confirmed by:
1. checking the DOD Award to Black & Veatch
CONT_IDV_HDTRA108D0007_9700, here:
https://t.co/G5ZEUhPm5U
2. click the tab for sub-awards
3. 7th down from top
4. 19-6192 is the sub-award ID
5. Note Date
3. Download the CSV file by clicking top right:
Again Data is confirmed (with more detail in Column 32 in the csv file)
4. CSV Details (Column 32)
Note Kiev, Ukraine Mentioned and Date 2019
5. CSV Details (2)
Mentions Labyrinth's HQ in Saint Petersburg, Florida, not Russia ;)
URL in CSV directs to original URL
https://t.co/G5ZEUhPm5U
Copy & Paste URL, not click.
Last column with date does not refute finding, it is:
"subaward_fsrs_report_last_modified_date"

More from Science
The physicist Hugh Everett III was born #OTD in 1930. His \u201crelative state\u201d formulation of quantum mechanics, which we now call the \u201cMany Worlds Interpretation,\u201d was published in 1957. pic.twitter.com/ZqMsZcPJDG
— Robert McNees, the bastegod (@mcnees) November 11, 2020
We look at the night sky and see photons arriving to us, emitted by distant stars. Let's contrast two different theories about how stars emit photons.
One theory says, we know how stars shine, and our equations predict that they emit photons roughly uniformly in all directions. Call this the "Many-Photons Interpretation" (MPI).
But! Others object. That is *so many photons*. Most of which we don't observe, and can't observe, since they're moving away at the speed of light. It's too ontologically extravagant to posit a huge number of unobservable things!
So they suggest a "Photon Collapse Interpretation." According to this theory, the photons emitted toward us actually exist. But photons that would be emitted in directions we will never observe simply collapse into utter non-existence.
If you are into cryptography or reverse engineering, you should love this.
Thread:

DNA consists of four different 'bases', A, C, G and T. These bases have specific meaning within our biology. Specifically, within the 'coding part' of a gene, a triplet of bases encodes for an amino acid

Most DNA is stored redundantly, in two connected strands. Wherever there is an A on one strand, you'll find a T on the other one. And similarly for C and G:
T G T C A G T
A C A G T C A
(note how the other strand is upside down - this matters!)

If you take all the DNA of an organism (both strands), you will find equal numbers of A's and T's, as well as equal numbers of C's and G's. This is true by definition.
This is called Chargaff's 1st parity rule.
https://t.co/jD4cMt0PJ0

Strangely enough, this rule also holds per strand! So even if you take away the redundancy, there are 99% equal numbers of A/T and C/G * on each strand *. And we don't really know why.
This is called Chargaff's 2nd parity rule.



















