Here's more on the new mitigation scenario for 1.5C. How does it work? What would society look like? Are we willing to do what's required to stop climate breakdown? See thread.
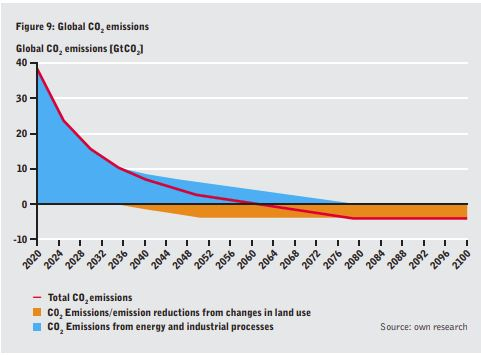
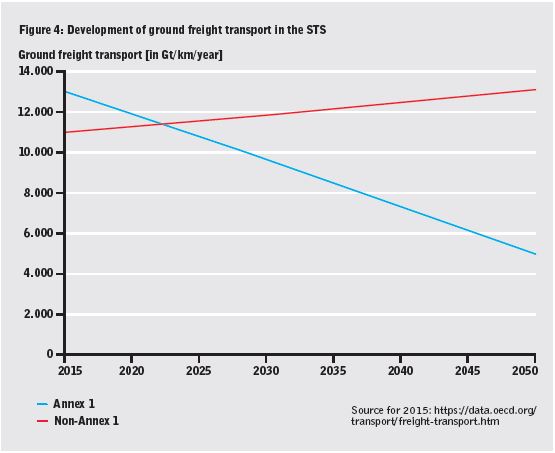
•a shift from private cars to public transportation
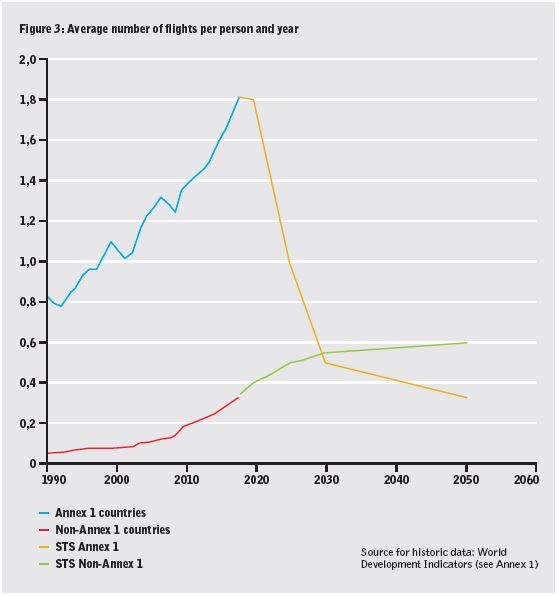
•reduction in flights
•smaller average house size
Meanwhile, consumption in the global South (non-Annex 1 countries) rises to converge.
•Meat consumption in high-income nations falls by about two-thirds (with specific focus on beef).
•Significant reduction in food waste.
•Global transition from industrial farming to regenerative agricultural methods to restore soils and biodiversity.
•Universal public services
•Shorter working hours
•Basic income and maximum wage
•Radical reduction in inequality
These measures ensure that all people have access to the resources they need to live flourishing lives even as aggregate economic output declines.
More from Science
Recently I learned something about DNA that blew my mind, and in this thread, I'll attempt to blow your mind as well. Behold: Chargaff's 2nd Parity Rule for DNA N-Grams.
If you are into cryptography or reverse engineering, you should love this.
Thread:

DNA consists of four different 'bases', A, C, G and T. These bases have specific meaning within our biology. Specifically, within the 'coding part' of a gene, a triplet of bases encodes for an amino acid

Most DNA is stored redundantly, in two connected strands. Wherever there is an A on one strand, you'll find a T on the other one. And similarly for C and G:
T G T C A G T
A C A G T C A
(note how the other strand is upside down - this matters!)

If you take all the DNA of an organism (both strands), you will find equal numbers of A's and T's, as well as equal numbers of C's and G's. This is true by definition.
This is called Chargaff's 1st parity rule.
https://t.co/jD4cMt0PJ0

Strangely enough, this rule also holds per strand! So even if you take away the redundancy, there are 99% equal numbers of A/T and C/G * on each strand *. And we don't really know why.
This is called Chargaff's 2nd parity rule.

If you are into cryptography or reverse engineering, you should love this.
Thread:

DNA consists of four different 'bases', A, C, G and T. These bases have specific meaning within our biology. Specifically, within the 'coding part' of a gene, a triplet of bases encodes for an amino acid

Most DNA is stored redundantly, in two connected strands. Wherever there is an A on one strand, you'll find a T on the other one. And similarly for C and G:
T G T C A G T
A C A G T C A
(note how the other strand is upside down - this matters!)

If you take all the DNA of an organism (both strands), you will find equal numbers of A's and T's, as well as equal numbers of C's and G's. This is true by definition.
This is called Chargaff's 1st parity rule.
https://t.co/jD4cMt0PJ0

Strangely enough, this rule also holds per strand! So even if you take away the redundancy, there are 99% equal numbers of A/T and C/G * on each strand *. And we don't really know why.
This is called Chargaff's 2nd parity rule.





















