ADHD, a thread (1)
If you have #ADHD & are struggling please know you’re not alone. This week on @Overview, we're talking about living w/ ADHD & Anxiety during the pandemic & many the stigmas & misconceptions that making things worse.
https://t.co/RO1ZGTlMq6


https://t.co/TXL9aOebDj
Like, right now, I’m furiously locked into this thread, & breaking away is super disconcerting & almost painful. When honed & controlled it can supercharge your productivity. But when unchecked it can feel like this.(12)

Quick warning, my choices and perspective don’t reflect a “right” or “wrong” way to do things. I believe everyone should do what works for best for them. (14)
https://t.co/PInu3lt8mi
https://t.co/1jJe4K02F9
https://t.co/7L6NJjSbA9
1. I want to talk about ADHD. I was diagnosed with ADD when I was 13-years-old. To say it's misunderstood (both by people who have ADHD and people who don't have it) is an understatement. What people need to understand about those of us who have ADHD is we are not homogeneous
— Yashar Ali \U0001f418 (@yashar) February 26, 2019
https://t.co/1jJe4K02F9
More from Health
The Health Protection (Coronavirus, Restrictions) (All Tiers and Self-Isolation) (England) (Amendment) Regulations 2021
£800 'house party' FPN & police can now access track & trace data
https://t.co/k9XCpVsXhC

“Large gathering offence”
As trailed by Home Secretary last week there is now a fixed penalty notice of £800 (or £400 if you pay within 14 days) for participating in an gathering of over 15 people in a private residence

Fixed Penalty Notices double for each subsequent “large gathering offence” up to £6,400
Compare:
- Ordinary fixed penalty notice is £200 or £100 if paid in 14 days
- Holding or being involved in the holding of a gathering of over 30 people is £10,000

Second big change:
Since September has been a legal requirement to sell-isolate if you test positive/notified by Track & Trace of exposure to someone else who tested positive
Police can now be given access to NHS Track & Trace data if for the purpose of enforcement/prosecution

This will make it easier for police to enforce people breaking self-isolation rules. Currently there has been practically no enforcement.
Data says only a small proportion of people meant to be self-isolating are fully doing so.
Very important that obvious failures with Track and Trace and self-isolation (study late last year said 18% of people complying https://t.co/dhJUZ7Pm0l) are not painted as an enforcement issue. Plainly not. Would just pass buck to police who have almost no capacity to enforce https://t.co/Eb4Kl5Ze0E
— Adam Wagner (@AdamWagner1) January 25, 2021
To start with, atheism is an unnatural self-contradicting doctrine.
Medical terminology proves that human beings are naturally pre-disposed to believe in God. Oxford scientists assert that people are "born believers".
https://t.co/kE0Fi588yn
https://t.co/OqyXcGIMJn

It should be known that atheism could never produce an intelligently-functioning society and neither ever will.
Contrastingly, Islam produced several intellectuals & polymaths, was on the forefront of scientific development, boasting 100% literacy
If the Muslim world had not existed, there literally would be no technology/achievements today.
— Starks\u262a\ufe0f\U0001f1f9\U0001f1e9 (@MegaIntelIect) January 8, 2021
Science only developed because of Islam, Europe should be grateful to Islam for civilizing their barbaric cult.
Source: The Caliph's Splendor, Pg 204-05 https://t.co/HVypO52Tpc pic.twitter.com/00jYSbaDSs
It is also scientifically proven that atheism led to lesser scientific curiosity and scientific frauds, which is also why atheists incline to pseudo-science.
Whereas, religion in general and Islam in particular boosted education.
https://t.co/19Onc84u3g

Atheists are also likely to affected by pervasive mental and developmental disorders like high-functioning autism.
Cognitive Scientists and renowned Neurologists found that more atheism is leads to greater autism.
https://t.co/zRjEyFoX3P

#FollowTheScience yes, but not just part of it!
THREAD👇
\U0001f534LIVE \U0001f4c5Today \u23f012:00 CET
— EU_HEALTH - #EUCancerPlan (@EU_Health) February 3, 2021
We are presenting today the #EUCancerPlan as part of a strong \U0001f1ea\U0001f1fa#HealthUnion
Follow the presentation live here: https://t.co/Cr8ATvzNkg#WorldCancerDay pic.twitter.com/zdByuklWV6
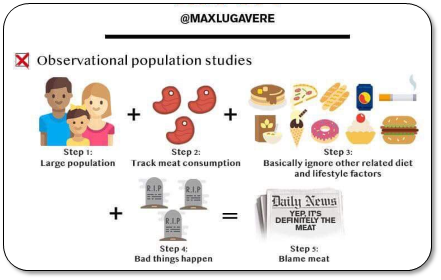
1/ Granted, some studies have pointed to ASSOCIATIONS of HIGH intake of red & processed meats with (slightly!) increased colorectal cancer incidence. Also, @WHO/IARC is often mentioned in support (usually hyperbolically so).
But, let’s have a closer look at all this! 🔍

2/ First, meat being “associated” with cancer is very different from stating that meat CAUSES cancer.
Unwarranted use of causal language is widespread in nutritional sciences, posing a systemic problem & undermining credibility.
3/ That’s because observational data are CONFOUNDED (even after statistical adjustment).
Healthy user bias is a major problem. Healthy middle classes are TOLD to eat less red meat (due to historical rather than rational reasons, cf link). So, they
4/ What’s captured here is sociology, not physiology.
Health-focused Westerners eat less red meat, whereas those who don’t adhere to dietary advice tend to have unhealthier lifestyles.
That tells us very little about meat AS SUCH being responsible for disease.
You May Also Like
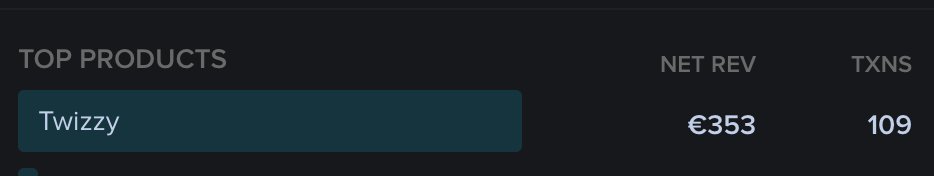
📈 ~12000 vistis
☑️ 109 transactions
💰 353€ profit (285 after tax)
I have spent 1.5 months on this app. You can make more $ in 2 days.
🤷♂️
I'm still happy that I launched a paid app bcs it involved extra work:
- backend for processing payments (+ permissions, webhooks, etc)
- integration with payment processor
- UI for license activation in Electron
- machine activation limit
- autoupdates
- mailgun emails
etc.
These things seemed super scary at first. I always thought it was way too much work and something would break. But I'm glad I persisted. So far the only problem I have is that mailgun is not delivering the license keys to certain domains like https://t.co/6Bqn0FUYXo etc. 👌
omg I just realized that me . com is an Apple domain, of course something wouldn't work with these dicks
As a dean of a major academic institution, I could not have said this. But I will now. Requiring such statements in applications for appointments and promotions is an affront to academic freedom, and diminishes the true value of diversity, equity of inclusion by trivializing it. https://t.co/NfcI5VLODi
— Jeffrey Flier (@jflier) November 10, 2018
We know that elite institutions like the one Flier was in (partial) charge of rely on irrelevant status markers like private school education, whiteness, legacy, and ability to charm an old white guy at an interview.
Harvard's discriminatory policies are becoming increasingly well known, across the political spectrum (see, e.g., the recent lawsuit on discrimination against East Asian applications.)
It's refreshing to hear a senior administrator admits to personally opposing policies that attempt to remedy these basic flaws. These are flaws that harm his institution's ability to do cutting-edge research and to serve the public.
Harvard is being eclipsed by institutions that have different ideas about how to run a 21st Century institution. Stanford, for one; the UC system; the "public Ivys".